FactoryTalk Design Studio: Transforming Industrial Automation Design
Tim Barthel, Executive VP at Cybertrol Engineering and former chairman of the Food Production Solutions Association (FPSA) Technology Network, shares his expertise in a "Tech Talk" highlighting a crucial aspect of food safety—the FDA's stringent requirements for validated information and how electronic reporting is revolutionizing compliance procedures. Watch the full video here.
Tradition has its place, but in the realm of data recording for food safety, traditional methods have proven to be cumbersome and error-prone. Circular chart recorders have long been a staple in the industry, gathering vital data points such as temperatures, flow rates, and chemical concentrations. These data points play an essential role in ensuring the safety of products throughout various stages of production, from ingredient handling to pasteurization.

However, the manual nature of these processes introduces room for human error and inefficiencies - especially when handwritten entries are involved. As ensuring food safety remains a top priority at Cybertrol Engineering, we’ll consider the transition from traditional circular chart recorders to cutting-edge electronic reporting systems, through the lens of CIP (Clean-in-Place) and SCADA (Supervisory Control and Data Acquisition) systems.
Electronic Reporting: A Paradigm Shift in Compliance Efforts
The emergence of electronic reporting has ushered in a new era of accuracy, efficiency, and data security in food safety compliance. Speaking from personal experience, even skeptical inspectors have become staunch advocates of the electronic transition. While many have long since appreciated the familiarity of traditional methods, especially in compliance with regulations like 21 CFR Part 11 and FDA requirements, the efficiency gains with newer electronic reporting are undeniable. We've extended this overhaul beyond the food and beverage sector, applying it to fermentation tank reports, cleaning processes, and more. Electronic reporting presents data in a linear, easily interpretable format, enabling swift identification of trends and anomalies. Additionally, real-time troubleshooting is facilitated through the display of operational steps, operator comments, and alarms.
The electronic reporting model replaces the paper-heavy filing cabinets with secure databases, granting authorized users access to versatile data formats. This facilitates rapid quality approvals and FDA compliance. Timestamped electronic signatures bring an added layer of validation, eliminating the ambiguities of handwritten signatures that can be susceptible to tampering.
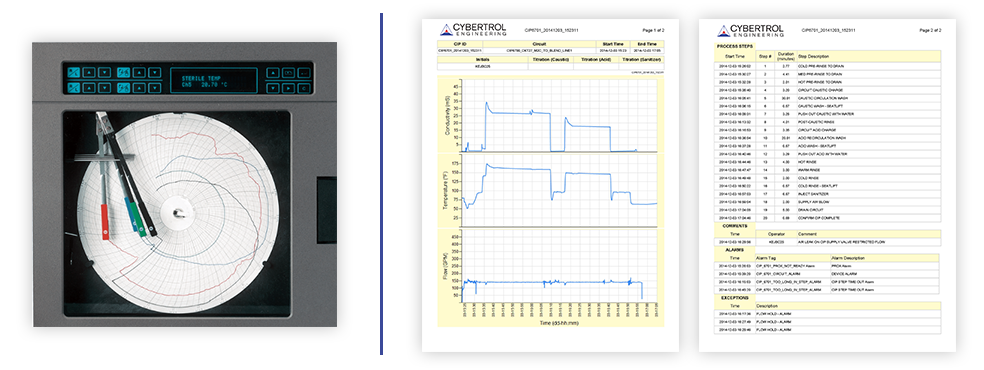 Image: Traditional CIP vs Modern Electronic CIP
Image: Traditional CIP vs Modern Electronic CIP
So, let's dissect the essence of this transformation. Both CIP and SCADA systems involve a control system, a PLC (programmable logic controller) on the factory floor, and field devices. However, the traditional setup connects these devices to a circular chart recorder, often ending up in a file cabinet for data storage and retrieval—a process that's akin to finding a needle in a haystack. In the electronic realm, the control system remains the same, but the data gets channeled into a secure database. This opens up a treasure trove of possibilities, allowing data to be harnessed in various formats accessible to authorized users. Think of it as an expansive window into your operations, streamlining quality approvals and regulatory compliance.
Overcoming Challenges and Future Directions
Of course, challenges exist. One such hurdle is pasteurization, where strict regulations curtail direct electronic reporting. However, innovative solutions are emerging, with digital chart recorders making waves, meeting even the stringent requirements of organizations like the PMO (Pasteurized Milk Ordinance). How does this change align with 21 CFR Part 11? Electronic reports boast time-stamped electronic signatures, a far cry from the scribbles of the past. Comments are integrated seamlessly, time-stamped and typed, accompanied by validated electronic signatures. Contrastingly, traditional CIP relied on handwritten notes and initials—an outdated practice.
Validation is another hurdle. Electronic systems offer transparent comparisons through the HMI (Human Machine Interface) gauge and historical data, a luxury the hardwired circular CIP chart recorder can't provide. The latter demands manual validation, comparing readings to physical gauges. Redundancy, a prized trait, finds a home in electronic reporting, enabling data backup, export, and even auto-generated PDF reports sent to designated recipients.
Last but certainly not least, security remains paramount. Electronic systems lock down access through Windows authentication, managed by the IT team, ensuring a robust and regulated environment. Off-site backups are automated, rendering the manual process of transporting physical records obsolete.
A Safer and More Efficient Future
Embracing electronic reporting demands a cohesive approach anchored in SCADA and CIP systems. These systems offer real-time monitoring, secure data storage, and user-friendly interfaces for operators. Robust security measures, including Windows authentication, version control, and access limitations, guarantee data accuracy and prevent unauthorized modifications.
This shift from traditional chart recorders to electronic reporting signifies a quantum leap in data management. It empowers industries with instant insights, streamlined compliance, and enhanced troubleshooting capabilities. The journey might be challenging, but it's a voyage well worth embarking upon. I, along with my team at Cybertrol Engineering, work hard to keep us at the forefront of system integration and automation, driving innovation to ensure food safety remains a top priority.
About Tim Barthel
Tim Barthel is the Executive Vice President at Cybertrol Engineering. With more than 30 years of industrial automation experience, he plays a key role in helping Cybertrol achieve its mission to create and deliver solutions to maximize manufacturing performance. Tim specializes in bringing a plantwide approach to every project with the goal of enabling system-wide visibility and control with common underlying infrastructure.

Cybertrol Engineering is a Supplier Member of the FPSA, as a provider of manufacturing solutions to food and beverage processors. Tim Barthel served as founding chair of the Technology Network from 2019-2024. In this role, he assumed responsibilities such as providing leadership to the Network, delegating responsibilities to accomplish the goals set in cooperation with the FPSA Staff Liaison, and reporting to the Networks' Leadership Team.
The mission of the Technology Network is to provide expertise, solutions, and guidance for FPSA member companies while creating an awareness of emerging trends within technology in food and beverage industries.
Ready to implement electronic reporting into your production process? Contact us today.
